

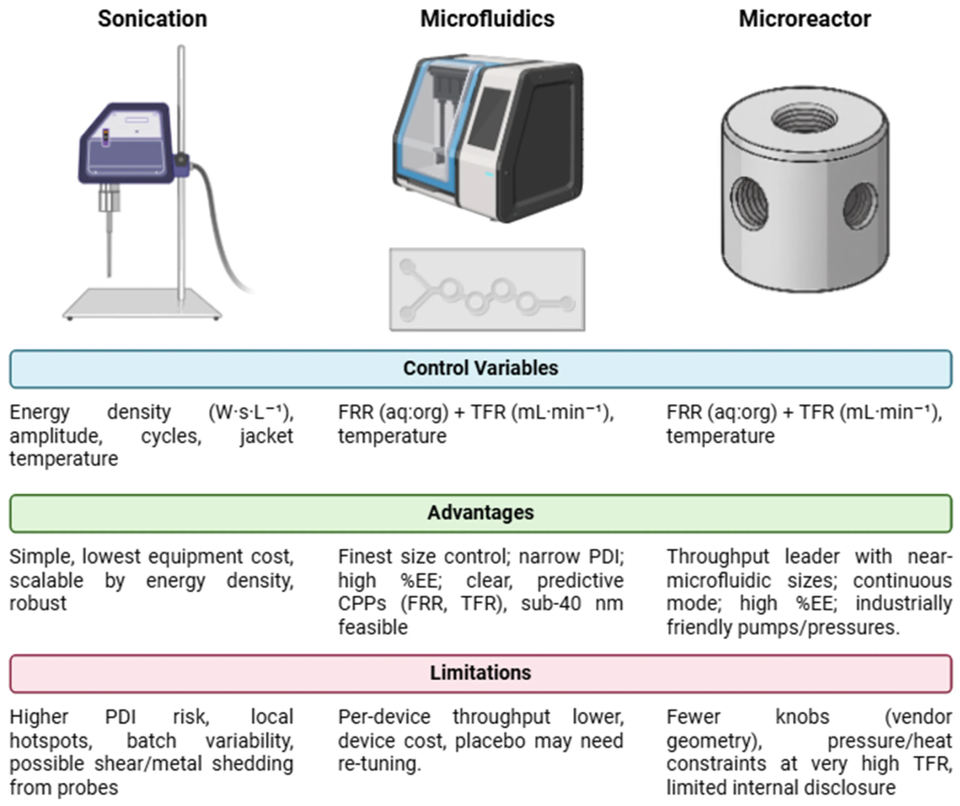
A recent publication arising from the ETERNAL Horizon Europe research collaboration compares three methods for fabricating diclofenac-loaded liposomes - probe sonication, microfluidic mixing, and a high-turbulence microreactor - under a Quality-by-Design framework.
The new peer reviewed paper, appearing in the journal MDPI Pharmaceutics, is the fruit of collaboration between ETERNAL researchers at Laboratorio Reig Jofre, MyBiotech GmbH, and IRIS Technology Solutions, along with research partners at the University of Barcelona and the Institute for Bioengineering of Catalonia. Entitled ‘Comparative Analysis of Sonication, Microfluidics, and High-Turbulence Microreactors for the Fabrication and Scaling-Up of Diclofenac-Loaded Liposomes’ the paper decribes how, having establsihed a processing-relevant liquid-crystalline temperature window of 70–85 °C for the lipid excipients via Differential Scanning Calorimetyr, a set of optimised conditions were established experimentally for the three methods under comparison.
Ultimately, all three routes met relevant Critical Quality Attributes (Average Particle Size ~50–100 nm, Poly-Dispersity Index ≤ 0.30, and high Encapsulation Efficiency) and the authors conclude that method selection should be guided by target size/dispersity and operational constraints: sonication enables energy-based scale-up, microfluidics offers precise size control, and microreactors provide higher throughput.
Read full open-access text at MDPI Pharmaceutrics now
This site uses cookies that enable us to make improvements, provide relevant content, and for analytics purposes. For more details, see our Cookie Policy. By clicking Accept, you consent to our use of cookies.