

A newly released technical report from ETERNAL's specialists in the field, presents a new environmental risk assessment (ERA) of existing pharmaceutical compounds, integrating monitoring data, emissions modelling, and multimedia fate modelling. The report identifies and ranks priority risks for freshwater environments and soils.
Pharmaceuticals are essential for modern healthcare, yet their widespread and continuous use results in the release of active pharmaceutical ingredients (APIs) into the environment. Following consumption and excretion, or through direct disposal, these substances enter wastewater systems and may subsequently reach surface waters, sediments, and soils (see diagram below).
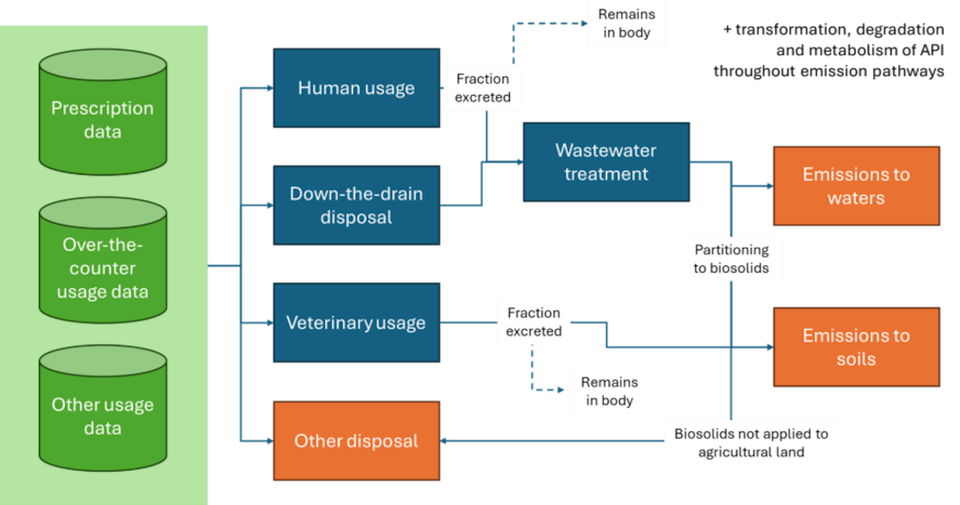
Pathways for APIs and associated transformation products from usage to emissions into surface waters and soils
While regulatory environmental risk assessments are routinely conducted for new medicinal products, many legacy or widely used existing pharmaceuticals were authorised before current environmental regulatory requirements were established. As a result, there remains a need to better characterise their environmental exposure and associated risks under real-world conditions.
The new report helps address this needs by presenting an environmental risk assessment of existing pharmaceutical compounds, integrating monitoring data, emissions modelling, and multimedia fate modelling. The work includes measured environmental concentrations (MECs) and predicted environmental concentrations (PECs) derived from spatially resolved emissions estimates and continental-scale fate modelling. By comparing empirical observations with modelled exposure estimates, the study evaluate the consistency / inconsistency between measurement- and model-based approaches, highlighting areas for future research.
The report builds on the basis of a previously formulated a list of priority in-use pharmaceuticals, based on risk ranking from monitoring and ecotoxicological data, previous risk ranking and prioritisation exercises and ensuring representation of different therapeutic classes. For a sub-set of selected priority substances, the report provides detailed results forming a spatio-temporal dashboard of the key indicators in the new assessment.
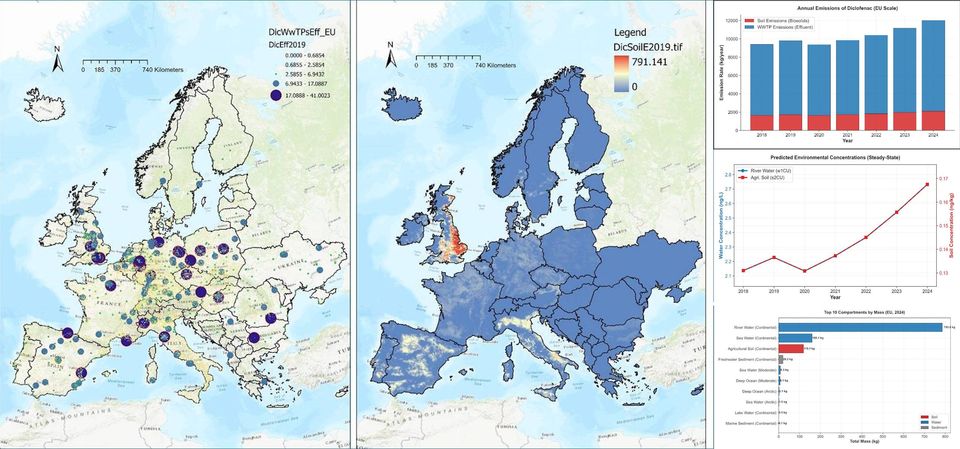
Spatio-temporal dashboard for a representative commonly used non-steroidal anti-inflammatory (Diclofenac)
The risk assessment is based on a collation of monitoring data across a broad selection of databases and literature, covering surface waters and soils. It compares these MECs to hazard values from a recently published library of species sensitivity distributions and other sources, alongside detection frequencies, to rank APIs in terms of those that present the highest risk to the environment. These results are compared to other risk ranking exercises, and placed in the context of PECs from the exposure modelling work. Finally, we use our findings to highlight priorities for future research towards better understanding of pharmaceuticals in the environment.
Download ‘Report on environmental risk assessment of existing pharmaceutical compounds’ now
This site uses cookies that enable us to make improvements, provide relevant content, and for analytics purposes. For more details, see our Cookie Policy. By clicking Accept, you consent to our use of cookies.